The scenario:
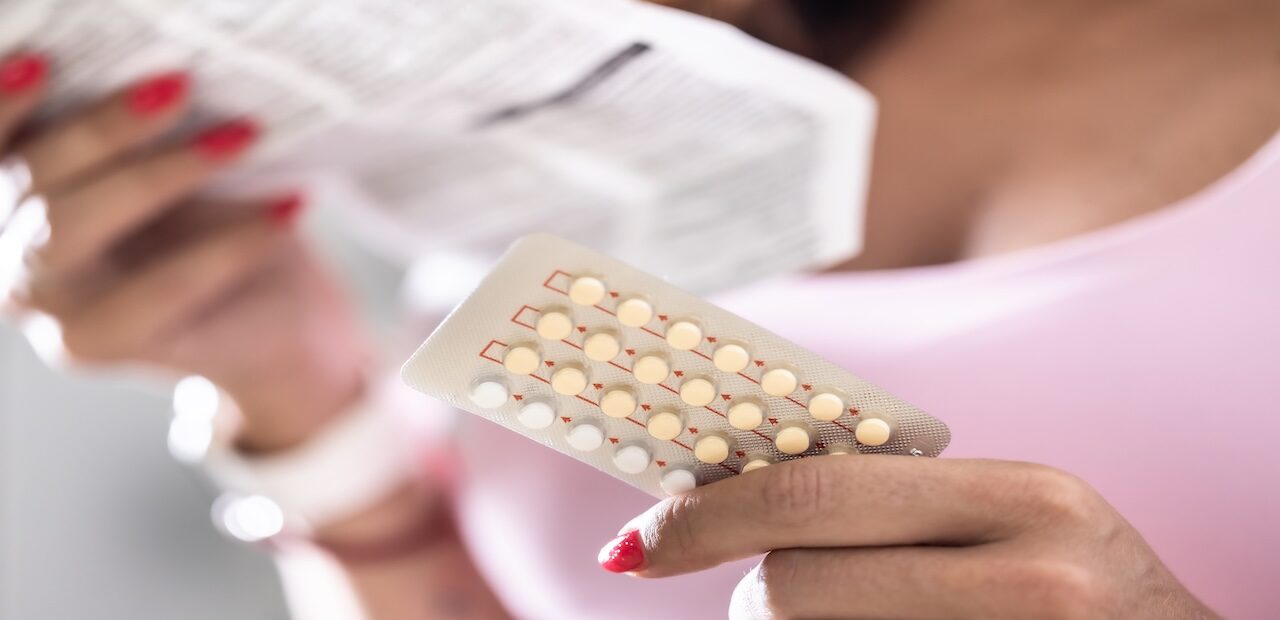
A 24-year-old patient (she/her) comes in after missing a period. She has a history of PCOS and irregular periods, but reports her periods have been fairly regular since taking birth control pills. In the clinic, her urine pregnancy test is positive. She is surprised and upset, as she’s been using a combined oral contraceptive (COC) for the past 2 years.
On further history, you learn she started a weekly GLP-1 injectable for weight loss about 6 months ago that she gets from a local med spa. She doesn’t remember the exact name but thinks it starts with a “s.” Since starting the injection, she’s had intermittent nausea and a few episodes of vomiting, especially in the days after injections and after she increases the dose. She asks “how did this happen?”